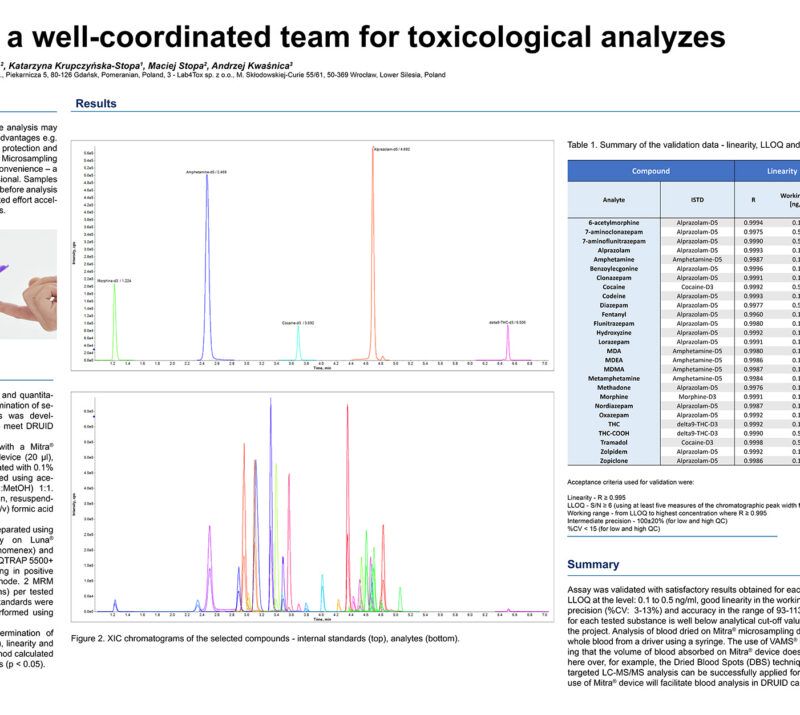
CardioCarePack – personalized medicine system for TDM of cardiological drugs based on LC-MS/MS analysis of samples collected at home with VAMS.Rafał Szewczyk1, 2; Adrianna Radulska3, Anna Lenartowicz2; Julia Mironenka2; Adrian Soboń1, 2; Katarzyna Krupczyńska-Stopa1, 2; Maciej Stopa1, 2; Tomasz Borkowski3; Ewelina Marciniak3, Leszek Kalinowski3 1Bioanalytic Sp. z o.o., Gdansk, Poland; 2LabExperts Sp. z o.o., Gdansk, Poland; 3 Medical University of Gdansk, Gdansk, Poland AbstractIntroductionCardiac arrhythmia affects approximately 12.6% of people over the age of 65. Ventricular arrhythmias are considered as responsible for 75% to 80% of sudden cardiac deaths. Drug therapy monitoring (TDM) in Anti-Arrhythmic Drugs (AAD) is essential for patient management when their have narrow therapeutic range, their use is associated with several serious adverse drug reactions, has a long, multiphasic elimination of up to several dozen days or formation of active metabolites with the action distinct form the parent drug. Developed assay named “CardioCarePack” is based on the use of capillary blood collected at home by a patient with volumetric absorptive microsampling (VAMS), quantitative analysis of selected drugs and their metabolites and a telemedical system that integrates all data between a doctor, patient and laboratory supporting the therapy process. Materials and MethodsThe project involved more than 300 patients who had been monitored for 2 years during regular pharmacological therapy. Patients were divided into 4 groups with the main anti-arrhythmic drug (Amiodaron, Propafenon, Sotalol and Digoxin), where additional ADD could also be administered. Every half a year during a visit in medical facility venous blood and capillary blood acquired by VAMS on 20 µl microsampling device (MITRA®) were collected. Between the visits patients were collecting samples by VAMS themselves at home. Samples were subjected to LC-MS/MS analysis QTRAP mass spectrometers (SCIEX). The LC-MS/MS method developed for 17 compounds covers therapeutic range of the tested compounds: 0.25 – 25 ng/ml (Digoxin and Nebivolol) 2.5 – 250 ng/ml (Metoprolol, Bisoprolol, Propafenone, Carvedilol, Perindopril, Ramipril, Spironolactone, Zofenopril and 25 – 2500 ng/ml (Sotalol, Desethylamiodarone, Eplerenone, Amiodarone, 5-hydroxypropafenone). ResultsOn the basis of samples collected during 4 visits in medical facility a correlation between drugs concentration in venous blood (serum) and capillary blood (MITRA®) (S/M) was calculated. For some compounds S/M ratio was close to 1 (ex. Sotalol – 0.95, %CV – 5.12%, Nebivolol – 1.03, %CV – 3.17%) or moderately to more than 2-fold different (ex. Digoxin – 0.69, %CV – 1.46%, Ramipril – 1.18, %CV – 11.1, Perindopril – 1.86, %CV – 4.73%, Amiodarone – 2.17, %CV – 10.78). The correlation factors are statistically significant (p < 0.05) and can be used for accurate concentration estimation in venous blood. Statistical analysis has also showed excellent correlation between two laboratories with the analysis on QTRAP 4500, 5500+, 6500+ mass spectrometers (Spearman correlations above R = 0.96). Processed data are placed in a server-based telemedical system where history, doses, therapeutic index flagging, cardiograms and other diagnostic results and data are available for patient, doctor and laboratory staff, respectively. Discussion and ConclusionsThe SARS-CoV-2 pandemic has forced changes in healthcare management because of the patients isolation and difficult access to the doctor. CardioCarePack helps in doctor’s supervision over the patient’s condition on the basis of collected within the software data and fits in with modern trends of home-based sample collection and personalized medicine. The project was co-financed by The National Centre for Research and Development and European Regional Develoment Fund. Grant no: POIR.01.01.01-00-1196/19 |

|


